Patient Safety and Risk Management
There is no such thing as zero risk. Every product has some form of risk for the patient or user of the product. For medical devices, the risks and impact on patient safety are most related to the product or the user of the device, while for pharma/biotech, the impact on patient safety to risk is confounded by the medicinal product, patient (i.e., genetics, allergies, tolerances, pre-existing conditions, etc.), and other medications.
Risk Management for medical devices starts with the initial development of the product, with risk tools understanding the potential hazards and failure modes that could affect the patient or user. ISO 14971 provides the guiding principles for developing a robust risk management and patient safety program. Over the course of development through manufacturing, the Design Control process helps identify and mitigate failures that pose a high risk to the patient or user. Bench testing easily identifies opportunities for improvement in the development of safe and effective Medical Devices.
Pharma/Biotech products have two challenges: Providing efficacy and safety; a balancing act between dosing, patient-to-patient variables, and disease management. For all the bench testing and pre-clinical studies performed to show efficacy, the need for patient testing eventually needs to show that the drug’s risk-benefit ratio is sufficient for drug approval. That task lies in the Drug Safety departments.
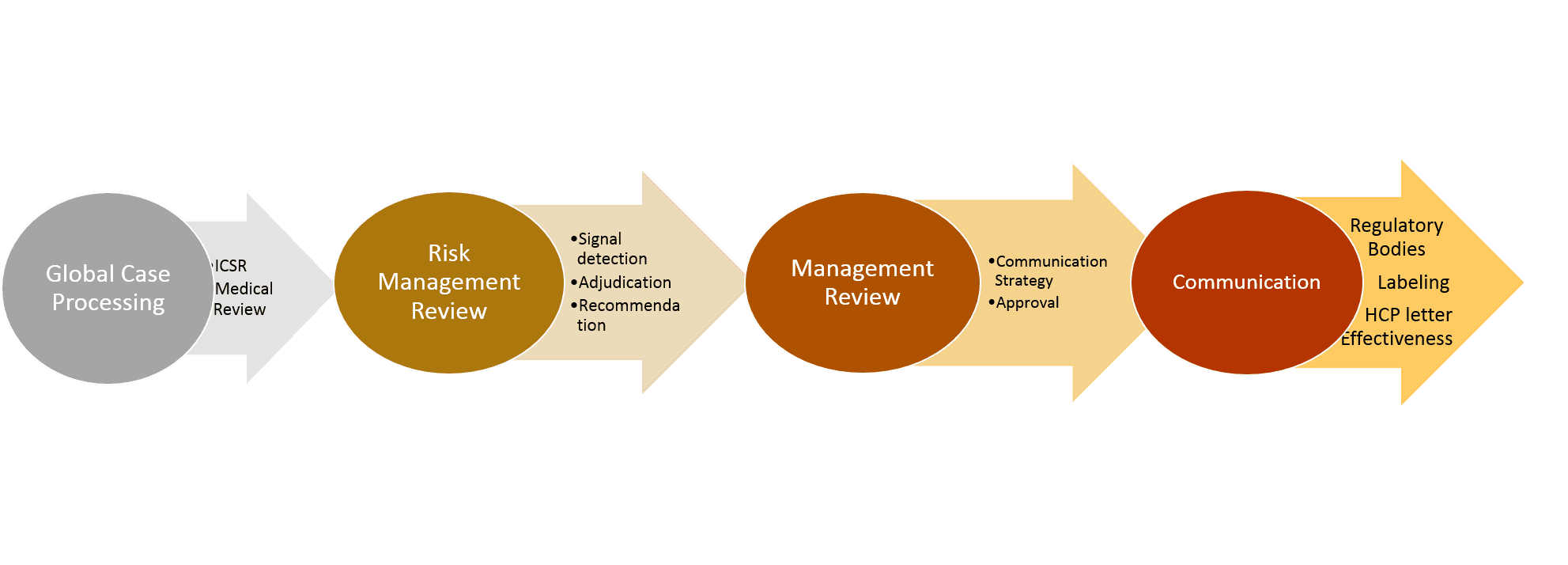
The detection of potential patient safety items is the same for devices and pharma… Setting up the correct process to identify, escalate, and communicate safety events:
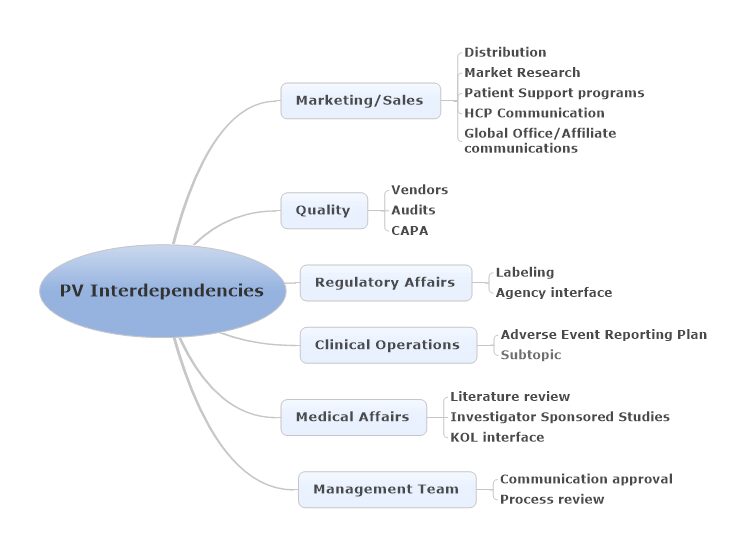
As Drug Safety transitions to a Quality System, as in the EUPV legislation, it depends on the interdependencies within the company:
Consult Wing has successfully implemented Device and Drug Safety programs throughout the healthcare spectrum. Helping companies execute global processes and tools for the sensitive detection of events, including investigation planning, communication, regulatory inspections, and developing risk communication tools (i.e., PSMF, Device Risk Files).
Contact us if you would like to discuss PV process enhancements or for us to perform an external audit to aid in continuous improvement opportunities.