Regulatory Compliance-Quality System
Exceptional Integrated Quality System: Enhancing the Efficiency and Efficacy of Your Services, Products, and Support.
Let us assist you in developing the optimal framework and programs to fulfill the fundamental components of a Quality System, whether it pertains to Medical Devices, Pharma/Biotech, or a Combination Product.
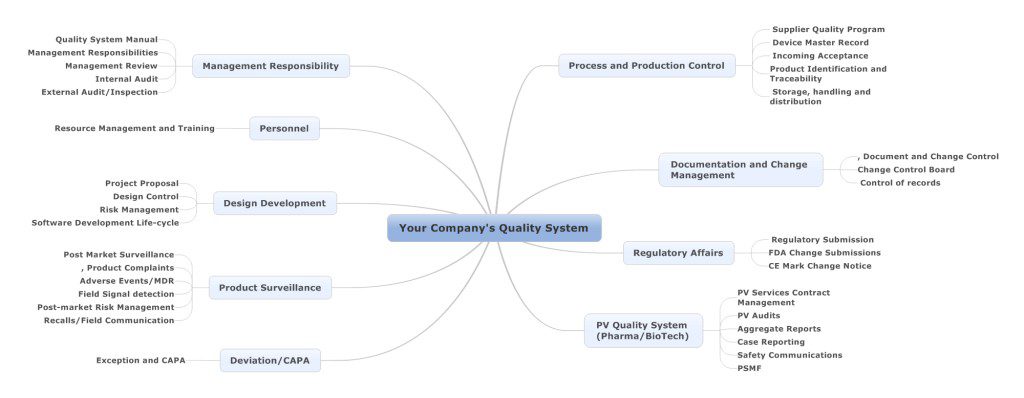
Quality Management System
Gain a comprehensive understanding of your organization's requirements and regulatory mandates, engage in short and long-term planning, develop an effective QMS, align it with your IT infrastructure, establish key performance indicators (KPIs) and metrics, and define management responsibilities.
Risk Management
From the initial concept through post-market activities, effectively manage the benefit-risk profile of your products. This encompasses designing the product with risk in mind, comprehending critical manufacturing processes to ensure product control, and developing sophisticated tools for post-market surveillance.
Deviation Management/CAPA
Adopt a risk-based approach to investigations, allowing for prioritization and consolidation of inquiries. This enables focused investigation into critical areas for your organization, leading to improved resource utilization and reduced cycle time for implementing corrections, corrective actions, and preventive actions.
Audit/Inspection Support
Evaluate your current organizational practices, prepare for regulatory inspections, provide support during inspections, and facilitate communication and remediation planning. With our extensive experience, we can help minimize inspection findings by aligning cross-functional processes and organizations to exceed regulatory compliance standards. Our aim is to ensure your organization is always inspection ready, whether it's preparing for a surprise visit or responding to a 3 AM phone call.
Supplier Quality
Starting from product development, we can assist you in qualifying suppliers, conducting audits, maintaining supplier listings, offering recall support, and facilitating management communication regarding critical vendor performance.

